|
|
|
|
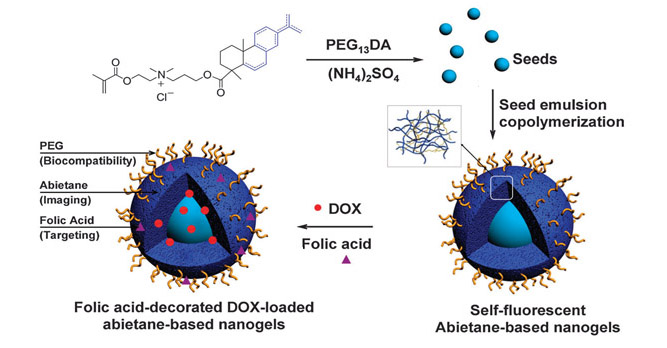
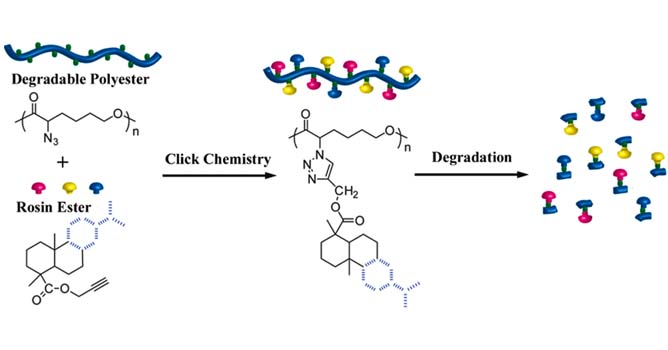
|
|
|
|
Research
|
|
|
Link to Google Scholar
I. Sustainable Biomass Chemistry and Biomaterials
Renewable Chemicals and Materials. Research in my laboratory has addressed a pressing global problem on sustainability. We have conceptualized the transformation of a class of hydrocarbon-rich biomass into sustainable chemicals, bioplastics, elastomers and biomaterials. The biomass includes resin acids, plant oils, fatty acids, and terpenes (Figure 1). Our goals are to develop these sustainable materials that are comparable to or even better than petroleum counterparts. We aim to discover chemistry that could lead to atom-efficient, eco-friendly and economy-feasible processes for converting biomass into useful intermediates, monomers and polymers. We also tackle some long-lasting hurdles in utilizing this class of biomass. We explored macromolecular approaches to making mechanically robust resin acid-containing polymers by enabling ultra-high molecular weight or by designing pentablock copolymers to surpass the low chain entanglement. We have capitalized on the transesterification and amidation to prepare plant oil-derived polymers with glass transition temperature spanning over one hundred degree. By further instilling some unique functional groups from biomass with robust efficient chemistry (e.g. reversible Diels-Alder, triazolinedione click Chemistry), we have been able to make high performance elastomers via grafting random copolymers, and shape-memory materials via dynamic and permanent networks. In addition, we have transformed nano-lignin and cellulose nanocrystals into renewable polymers to achieve tough bioplastics.
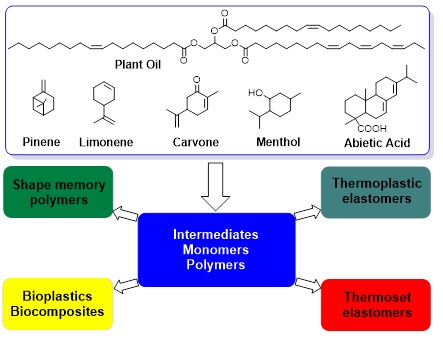
Figure 1. Selected biomass and their polymeric targets.
Discovery of Natural Product Antimicrobials. We are interested in discovering new antimicrobial agents to kill dru-resistant superbugs ( Figure 2). Another direction is to design new antimicrobial agents from hydrocarbon-rich natural products. We have found that natural resin acid-derived cationic compounds and polymers can be used as very effective antimicrobial materials to kill both Gram-positive and Gram-negative bacteria while maintaining low cytotoxicity to mammalian cells. Both molecular dynamics simulation and model lipid dye-leakage experiments confirm that natural resin acid-derived cationic compounds first attach to negative charged membranes through electrostatic interactions, followed by hydrophobic interactions between hydrophenanthrene and lipid tails, leading to membrane leakage and disruption.
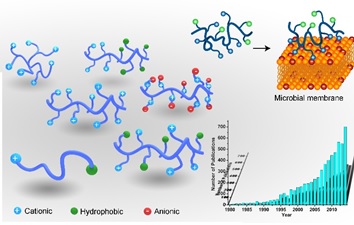
Figure 2. Macromolecular antimicrobials using natural products.
Selected Publications:
- Wang J.; Yuan L.; Wang Z.; Rahman M. A.; Huang Y.; Zhu T.; Wang R.; Cheng J.; Wang C.; ChuF.*; Tang C.* Photoinduced Metal-Free Atom Transfer Radical Polymerization of Biomass Based Monomers. Macromolecules, 2016, 49, 7709-7717.
- Ganewatta M. S,; Ding W.; Rahman M. A.; Yuan L.;, Wang Z.; Hamidi N.; Robertson M. L.*; Tang C.* Biobased Plastics and Elastomers from Renewable Rosin via “Living” Ring-Opening Metathesis Polymerization, Macromolecules, 2016, 49, 7155-7164.
- Wang Z. K.; Zhang Y.; Yuan L.; Hayat J.; Trenor N. M.; Lamm M.; Vlaminck L.; Billiet S.; Du Prez F. E.; Wang Z. G.; Tang C.* Biomass Approach toward Robust, Sustainable Multiple Shape-Memory Materials, ACS Macro Lett. 2016, 5, 602–606.
- Ganewatta M. S.; Miller K. P.; Singleton S. P.; Mehrpouya P.; Chen Y.-P.; Yan Y.; Nagarkatti M.; Nagarkatti P.; Decho A. W.;* Tang C.* Bacterial Biofilm Disrupting Sustainable Antimicrobial Coatings from Resin Acid Derived Materials. Biomacromolecules, 2015, 16, 3336–3344.
- Ganewatta, M. S.; Tang C.* Controlling Macromolecular Structures towards Effective Antimicrobial Polymers, Polymer, 2015, 63, A1-A29.
- Yuan L.; Wang Z.; Trenor N. M.; Tang C.* Robust Amidation Transformation of Plant Oils into Fatty Derivatives for Sustainable Monomers and Polymers, Macromolecules, 2015, 48, 1320-1328.
- Ganewatta M. S.; Chen Y.-P.; Wang J.; Zhou J.; Ebalunde J.; Nagarkatti M.; Decho A. W.*; Tang C.* Bio-inspired Resin Acid-Derived Materials as Anti-Bacterial Resistance Agents with Unexpected Activities. Chem. Sci., 2014, 5, 2011 - 2016.
- Yao K.; Tang C.* Controlled Polymerization of Next-Generation Renewable Monomers and Beyond. Macromolecules (Perspective) 2013, 46, 1689-1712.
|
|
|
II: Organometallics and Metallopolymers
Synthetic Methodology on Cationic Metallocene Derivatives and Polymers. We have led the accomplishment of a new platform of organometallic and metallopolymer chemistry based on
cationic metallocenes (or metallocenium). We developed synthetic strategies to prepare metallocenium derivatives: nucleophilic addition and hydride abstraction ( Figure 3). These reactions are highly dictated by the steric effect and electron-donating ability of nuceophiles as well as oxidation reagents for hydride abstraction. Systematic methdologies on the preparation of various monomers and controlled polymerization toward side-chain metallocenium-containing polymers have been established.
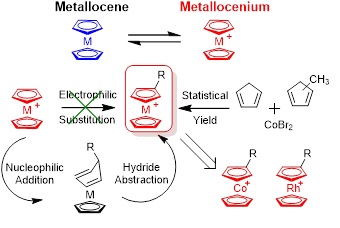
Figure 3. Synthesis of cationic metallocene (metallocenium) derivatives.
Polyelectrolyte Chemistry. Metallocenium polymers are a new class of polyelectrolytes, as the cations are metal centers in the polymers, which are significantly different from conventional quaternary ammonium- and phosphonium-based polyelectrolytes. We have studied both quantitative and qualitative ion exchange processes with a variety of anionic species. Counterion-induced self-assembly has been utilized to produce an array of intriguing nanoscale morphology from amphiphilic copolymers.
Metallopolymer-Antibiotic Bioconjugate Antimicrobials. As a new class of cationic polyelectrolytes, metallocenium-containing polymers have enormous potential for biomedical applications. We have taken efforts in exploring medicinal chemistry of cationic metallocene derivatives and polymers. The advantage of the use of cobaltocenium is its stability in the blood circulation, while ferrocenium is susceptible for quick decomposition. We have created a new paradigm of these metallopolymers to reinstate vitality of antibiotics against multidrug resistant bacteria by proactively disabling bacterial defense systems (Figure 4). We have demonstrated that the ability of ion-exchange for these polymers plays a key role in inhibiting the activity of β-lactamase enzyme that is commonly used by bacteria to hydrolyze antiobiotics such as penicillin and amoxicillin. In addition, these cationic metallopolymers could directly interact with negatively charged bacterial cell membranes to kill bacteria. These findings could pave a new platform for the design of antibiotics and antimicrobial agents to battle multidrug resistant bacteria and superbugs.
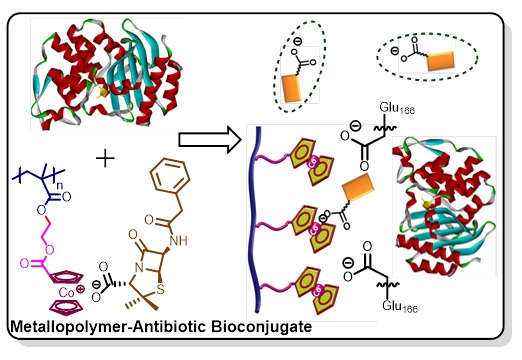
Figure 4. Antimicrobial metallopolymer-antibiotic bioconjugates.
Selected Publications:
- Yang P.; Pageni P.; Kabir P.; Zhu T.; Tang C.* Metallocene-containing Homopolymers and Heterobimetallic Block Copolymers via Photoinduced RAFT Polymerization. ACS Macro Lett. 2016, 5, 1293-1300.
- Yan Y.; Zhang J.; Ren L.; Tang C.* Metal-Containing and Related Polymers for Biomedical Applications. Chem. Soc. Rev. 2016, 45, 5232-5263.
- Zhang J.; Yan J.; Pageni P.; Yan Y.; Wirth A.; Chen Y-P.; Qiao Y.; Wang Q.; Decho A. W.; Tang C.* Anion-Responsive Metallopolymer Hydrogels for Healthcare Applications, Sci. Rep. 2015, 5, 11914.
- Yan Y.; Deaton T.; Zhang J.; He H.; Hayat J.; Pageni P.; Matyjaszewski K.; Tang C.* Syntheses of Monosubstituted Rhodocenium Derivatives, Monomers and Polymers, Macromolecules 2015, 48, 1644-1650.
- Zhang J.; Chen Y.-P.; Miller K. P.; Ganewatta M. S. Bam M.; Yan Y.; Nagarkatti M.; Decho A. W.; Tang C.* Antimicrobial Metallopolymers and Their Bioconjugates with Antibiotics against Multidrug Resistant Bacteria, J. Am. Chem. Soc. 2014, 136, 4873-4876.
- Hardy C. G.; Zhang J.; Yan Y.; Ren L.; Tang C.* Metallopolymers with Transition Metal in Side Chain by Living and Controlled Polymerization Techniques, Prog. Polym. Sci. 2014, 39, 1742-1796.
- Zhang J.; Yan Y.; Chance W. M.; Chen J.; Hayat J.; Ma S.; Tang C.* Charged Metallopolymers as Universal Precursors for Versatile Cobalt Materials, Angew. Chem. Int. Ed. 2013, 52, 13387 –13391.
|
|
|
III. Polymers for Dielectric Energy Storage
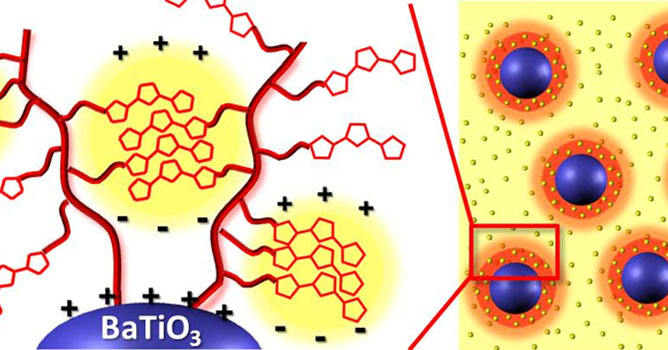
We invest a small effort toward novel dielectric polymers. We have discovered a class of thiophene-containing polymers as high performance dielectric materials for energy storage. These polymers are significantly different from conventional dielectric polyvinylidene fluoride and polypropylene. These polymers can form ultra-small polarizable nanocrystalline domains embedded in an insulating matrix, leading to high dielectric permittivity, while maintaining low dielectric loss. Our recent efforts have led to novel nanocomposites based on thiophene polymer-grafted barium titanate nanoparticles that exhibit unprecedented dielectric properties ( Figure 5). We have designed monomodal and biomodal polymer brushes from the BT nanoparticles via RAFT polymerization.
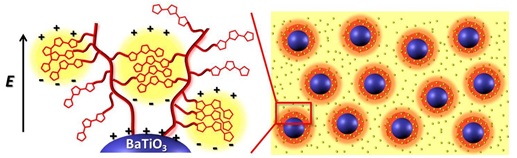
Figure 5. Thiophene polymer-grafted barium titanate as dielectric nanocomposites.
Selected Publications:
- Qiao Y.; Yin X.; Wang L.; Islam M. S.; Benicewicz B.; Ploehn H. J.; Tang C.* Bimodal Polymer-Brush Core-Shell Barium Titanate Nanoparticles: A Strategy for High-Permittivity Polymer Nanocomposites, Macromolecules, 2015, 48, 8998–9006.
- Qiao Y.; Islam M. S.; Wang L.; Yan Y.; Zhang J.; Benicewicz B. C.; Ploehn H. J.*; Tang C.* Thiophene Polymer-Grafted Barium Titanate Nanoparticles toward Nanodielectric Composites. Chem. Mater. 2014, 26, 5319-5326.
- Qiao Y.; Islam M. D.; Han G.; Leonhardt E.; Zhang J.; Wang Q.; Ploehn H. J.; Tang C.* Polymers Containing Highly Polarizable Conjugated Side Chains as High Performance All-Organic Nanodielectric Materials, Adv. Funct. Mater., 2013, 23, 5638–5646.
|
|
|







